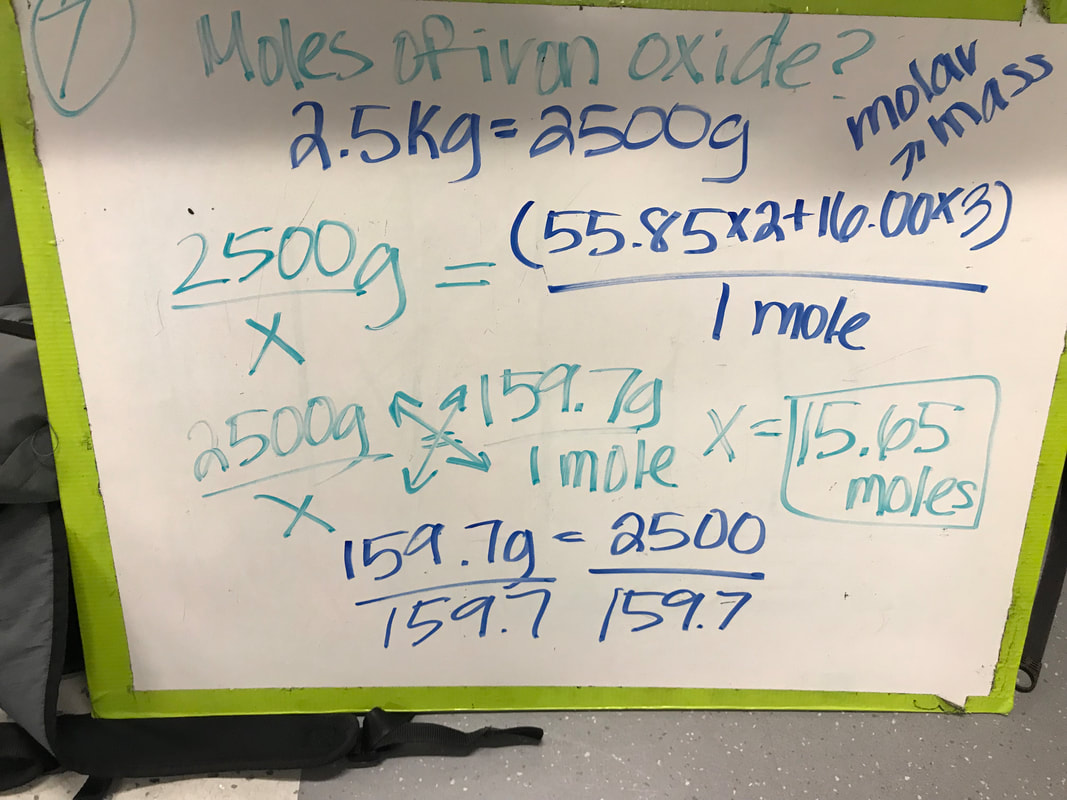Unit 4: Mass Ratios
Unit 4 Review Board:
Big Ideas:
|
Cross Cutting Concepts:
|
Scale, Proportion & Quantity: In Unit 4, we learned that moles can be used to compare different elements because the particles of elements vary in size, so you cannot use mass as a comparison, but you can use the number of particles. We used proportions and cross multiplication to convert between grams and moles or vice versa. We also discovered how to find the empirical formula of a compound by use cross multiplication or dividing by the molar mass to convert grams to moles and then dividing by the smaller number of moles to get the ratio of elements in a compound. Structure and Functions: In this unit, we used legos to model different types of compounds to see how certain elements bond with each other. In order to create a stable compound, the legos had to fill in two rows with no gaps. This meant that for a molecule to be stable, each atom must use its full bonding capacity. As we saw with our example of Ethanol, the legos could be arranged in two different ways, creating either Ethanol or Dimethyl ether. These two compounds had the same molecular formulas, but their properties were completely different. This led us to conclude that structure gives a substance its properties. Patterns: Throughout this unit, we struggled to find a pattern to explain why some compounds exist and others do not. However, after making the lego models, we learned that elements can only bond with so many other elements without there being gaps, making the compound unstable. We also observed that the hydrides of the elements in Mendeleev's table seemed to go up by one hydrogen and continue down the column of elements. We predicted that the last row would either bond with 0 hydrogens or 2 hydrogens, so that it would continue the pattern. |